Seaweed is causing havoc in our oceans. Together, scientists from Mexico and the UK are working hard to find inventive solutions to the seaweed which washes onto shores every summer in the Caribbean.
Since 2011, a huge raft of free-floating seaweed has been growing every summer in the Atlantic Ocean. The seaweed, called Sargassum, is a macroalgae, a large, brown, open ocean species which can be identified by small air sacs which help it to float.
The seaweed grows quickly, creating large floating clumps in the ‘Sargasso Sea,’ a region of the Atlantic Ocean where multiple currents meet. Currents carry these clumps south. Favorable conditions cause it to grow out of control. The largest annual raft is named the Great Atlantic Sargassum Belt (GASB). It stretches from Mexico to the west coast of Africa, it is over 8500km in length. In 2018, the GASB was made up of over 20 million tonnes of biomass!
The vast increase in growth every year is thought to be caused by fertiliser run-off from farmland in North America and the Amazon basin, as well as seasonal upwelling from the deep sea off Africa’s west coast, bringing nutrients to the area. Changes in the climate and ocean currents are also making the conditions perfect for Sargassum to grow quickly. Studies show that in the last 40 years, the sea surface temperature has increased by 1˚C and the summer has extended by almost a month. It is feared that the large blooms of seaweed leave behind seeding patches which can survive through the winter and restart the bloom the following summer.
We can observe the Sargassum blooms using satellites. This species has a chlorophyll pigment which reflects infrared light more strongly than the surrounding seawater. To satellites which detect infrared, Sargassum looks like a blazing fire and can be mapped.
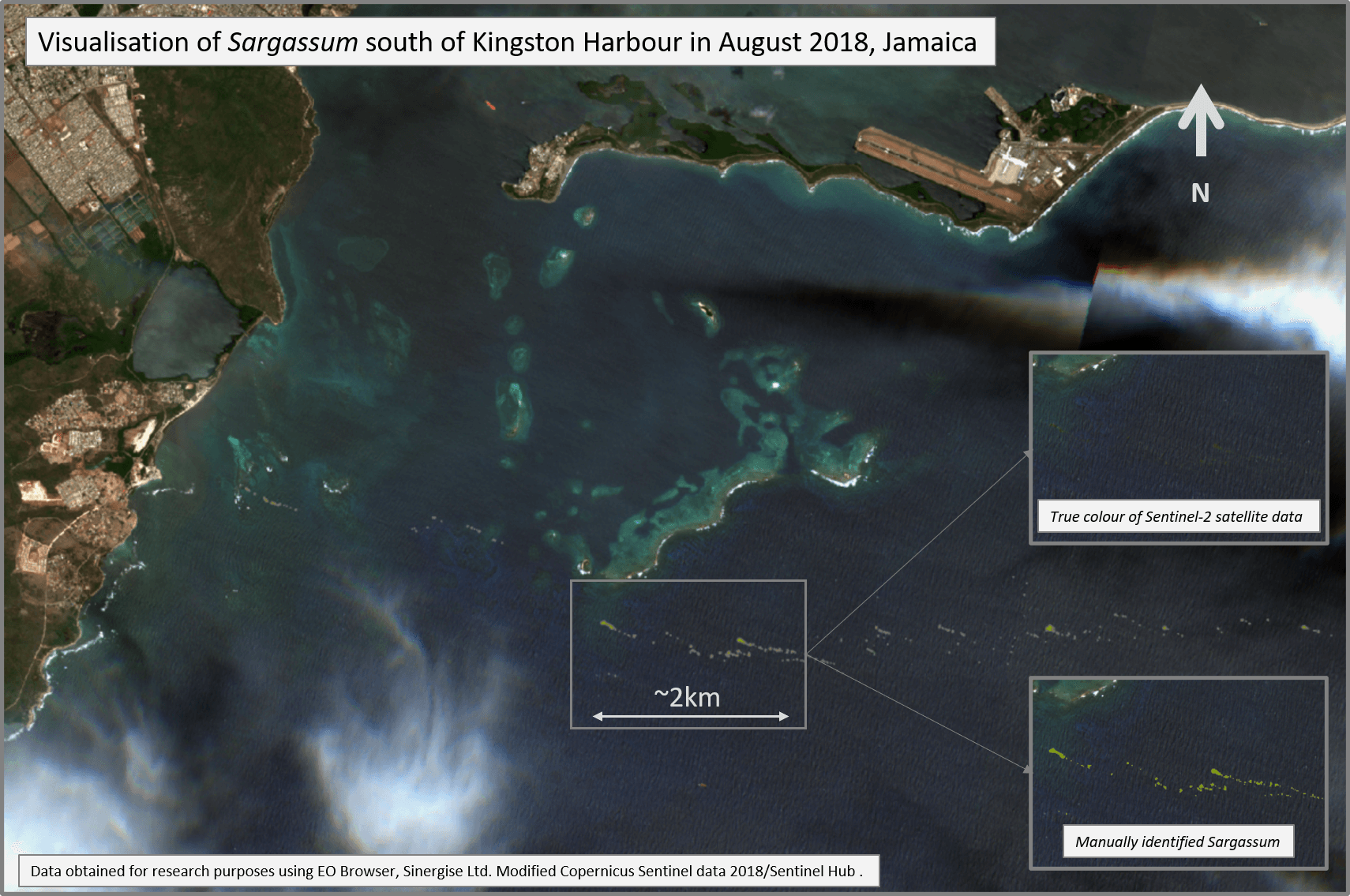
Satellite visualisation of Sargassum. Credit: Sien Van Der Plank, SARTRAC
At sea, the seaweed mats provide havens for many marine species and create ocean ecosystems. In fact, there is even a Sargassum fish which lives in the seaweed, changing colour to camouflage with its surroundings. The frogfish species can survive out of the water on top of the mats for some time when threatened. Other fish and marine turtle species use the raft for shelter and nursery grounds. Reports have also identified over 30 species of cetacean either resting or feeding in the Sargasso Sea. The area is often referred to as the ‘golden jungle,’ because of the large number of species. However, once washed ashore in excessive amounts, the seaweed can have the opposite effect, suffocating coral reefs, seagrass beds and turtle nesting sites, as well as having a long-term impact on the tourism industry. In 2018, there was an estimated $3.5 billion loss due to the lack of tourists visiting Mexico alone because of the problems associated with rotting seaweed on the beach. Millions of dollars are also spent each year collecting and removing the seaweed from the beach. Some countries have set up nets at sea to try and block the incoming seaweed, but it does not work.

Sargassum on the Mexican shoreline. Credit: Rebecca Dowell
Scientists all over the world are working to improve the tracking of Sargassum blooms, preventing it reaching the shore and researching sustainable processes which can use the seaweed that washes up on beaches as a useful resource. So far, people have used it to make shoes, stationary and even bricks, which have been used to build local houses.
PhycoMExUK is a group of researchers from universities, scientific organisations and industrial partners in both the UK and Mexico who have teamed up to tackle the crisis using new processing techniques to turn the seaweed into useful products. The process is called Hydrothermal liquefaction (HTL) and uses high temperatures (about 350˚C) and pressures (about 200 bar – the same as the pressure at 2500m deep below the sea) The HTL process takes place in an enclosed bioreactor. It breaks down the seaweed into other products. After 15-30 minutes, the reaction is complete and four different fractions are collected. The carbon dioxide gas is released and can be used to re-heat the reaction system.
The remaining solution is filtered, and a solid powder often referred to as char is collected. This char contains lots of carbon and is also where all the toxic heavy metals, which are absorbed by the seaweed, end up. The char powder can be used to return carbon to soils and provide extraction opportunities for heavy metals. The remaining liquid is separated into two fractions: bio-crude and aqueous. The aqueous fraction contains lots of ammonia and phosphate so can be used as a natural fertiliser for crops in the local agriculture industry. The bio-crude is treated and then can be used in the production of biofuels.

The HTL process. Credit: Amy Pilsbury
The process is beneficial because it can cope with wet and salty biomass so there is no need for expensive and time-consuming washing and drying processes. Perhaps even better, the process can deal with plastic pollution, removing it from the ocean and recycling it into products along with the seaweed.
Changing the temperature, pressure and reaction time changes the amount of each fraction which is produced. The group are working to find the perfect reaction conditions to produce the highest yield of each product so that the conditions can be changed to meet product demand. The aim of the project is to create a sustainable biorefinery which can collect and process the seaweed straight from the sea before it reaches the shore which will protect inshore ecosystems and encourage tourists to return to the Caribbean regions easing the economic impact of the seaweed influx.
Download PDF
If you wish to save, or print, this article please use this pdf version »


